Pancreatic cancer has long stood apart as a diagnosis doctors dread to pronounce and patients rarely survive, yet a Spanish research team has now reported results that challenge this bleak script in a striking way.
A cancer that rarely gives patients a second chance
Pancreatic cancer is often described by oncologists as a “silent sprinter”. It grows quietly, spreads fast, and is usually picked up only when the disease has already advanced. At that stage, surgery helps only a minority of patients and chemotherapy buys limited time.
In countries across Europe and North America, five-year survival still hovers around single digits. Many patients die within the first year after diagnosis. Late detection, aggressive biology and limited treatment options combine into a grim equation.
One gene, called KRAS, sits at the heart of this malignancy. It is mutated in around 90% of pancreatic tumours. KRAS acts like a jammed accelerator pedal, keeping growth signals permanently switched on. Drug companies have chased it for decades, with only modest success.
Some newer medicines can now hit certain KRAS mutations. In pancreatic cancer, though, these drugs have so far only managed to slow tumour growth for a few months. The cancer almost always finds a way around the blockade.
For years, KRAS inhibitors offered short pauses, not lasting control, as pancreatic tumours quickly learned to bypass the single target.
That frustrating pattern pushed scientists at Spain’s National Cancer Research Centre (CNIO) in Madrid to rethink the whole strategy. If hitting one target wasn’t enough, could hitting several at once change the game?
A triple therapy designed to block cancer on three fronts
The team, led by veteran cancer researcher Professor Mariano Barbacid, decided to test a high-risk idea in mice engineered to develop pancreatic cancer. Instead of relying on one drug, they combined three, each acting on a different stage of the same growth pathway.
The backbone of the mix was an experimental KRAS inhibitor called daraxonrasib. Added to it was afatinib, a medicine already approved for certain lung cancers that blocks so‑called ErbB receptors on the cell surface. The third piece was SD36, a molecule that tags specific proteins for destruction inside the cell.
➡️ Experte schlägt Alarm bei diesem Flaschenwasser: „Es sollte verboten werden“
➡️ Diese einfache Gewohnheit vor dem Schlafengehen hilft dem Gehirn abzuschalten
➡️ Diese Technik hilft, Gedanken abends schneller loszulassen
➡️ Wie Sie mit einem DIY-Regal für Parfums das Badezimmer luxuriöser und funktional gestalten
These three compounds are designed to interfere with a chain of molecular events that KRAS normally controls:
- Daraxonrasib tries to switch off the mutant KRAS engine at its source.
- Afatinib cuts down some of the incoming growth signals that feed into KRAS pathways.
- SD36 clears out key proteins further downstream, choking off the messages that tell cells to divide.
By attacking at several points, the researchers hoped to make it much harder for tumour cells to adapt or reroute their signalling.
The Spanish team likens the strategy to suspending a bridge from three cables instead of one: damage one, and the other two still hold everything in place.
What happened when the trio was tested in mice
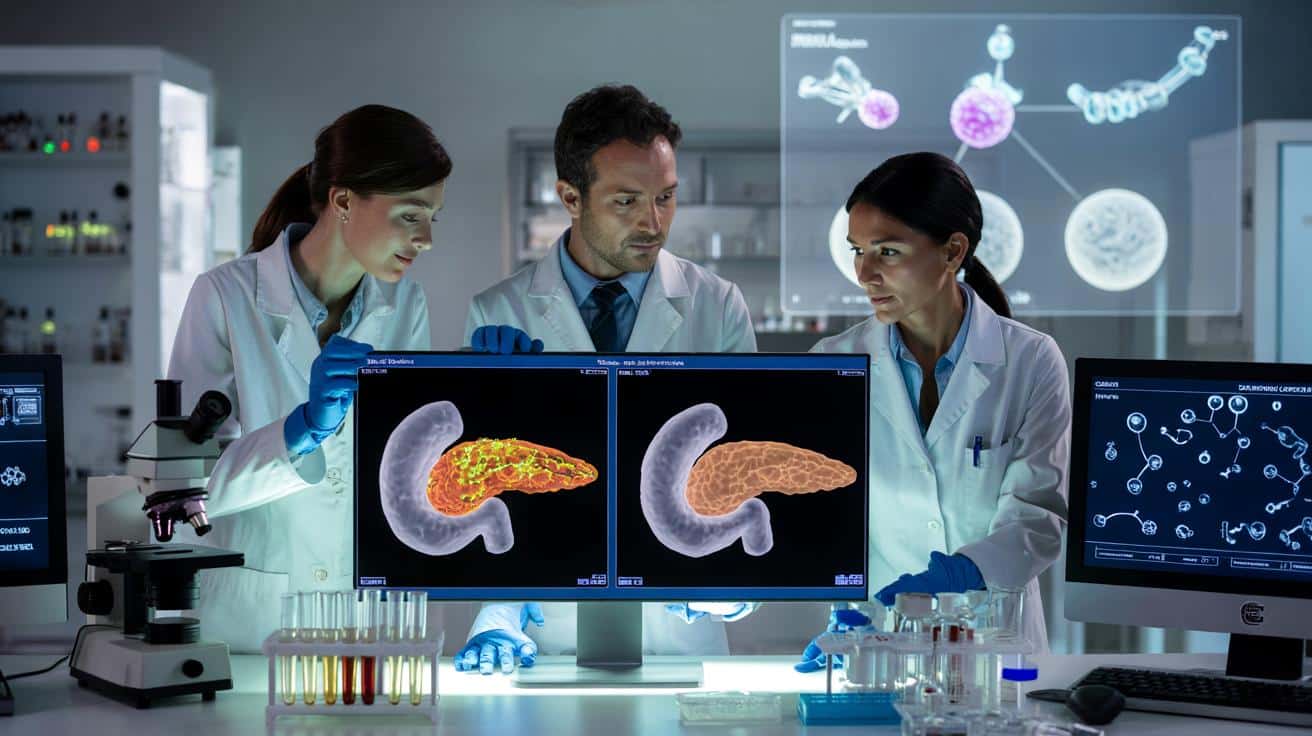
The results, published in the journal PNAS, were unexpected even for the researchers. In three separate mouse models of pancreatic cancer, the triple combination led to a complete disappearance of tumours on imaging tests.
Equally striking, the tumours did not return after treatment stopped. In most previous studies with single or double drug regimens, cancers grew back once therapy ended, sometimes more aggressively than before.
Here, the prolonged absence of regrowth suggests that the cancer cells had run out of escape routes. The multi-level blockade may have been tight enough, and sustained long enough, to prevent the usual wave of new resistance mutations.
Another key finding: the mice tolerated the treatment surprisingly well. Researchers reported no serious toxic effects or organ damage during the study period, although more detailed safety work still needs to be done.
| Component | Main role in the combo | Current status |
|---|---|---|
| Daraxonrasib | Directly targets mutant KRAS | Experimental, not yet approved |
| Afatinib | Blocks ErbB receptors feeding growth signals | Approved for some lung cancers |
| SD36 | Marks specific proteins for destruction | Experimental research tool |
In mice, the three-drug attack wiped out pancreatic tumours and kept them from coming back, without major toxicity reported.
Cautious optimism: from mouse cages to hospital wards
Despite the excitement, the Madrid team is stressing caution. Cancer drugs are notorious for behaving very differently in humans than in rodents. Many treatments that looked brilliant in mice failed later in patients.
Adapting this triple therapy for people will mean juggling several delicate variables. Dosing schedules must be refined so that the three drugs reinforce each other without overwhelming healthy tissues. Long-term side effects need careful tracking. Potential interactions with standard chemotherapy also have to be mapped.
Professor Barbacid has said that more preclinical work is required before the team can even propose a first-in-human trial. That planning phase alone may take years, as each component of the combo must pass regulatory checks and toxicology testing.
Why this approach matters beyond pancreatic cancer
Even with those caveats, oncologists are paying close attention. The CNIO study does more than showcase three specific molecules. It supports a broader principle: multi-target strategies may be key for beating cancers that quickly outwit single drugs.
Pancreatic tumours are not the only ones driven by KRAS. Some lung and colorectal cancers rely on the same pathway, and they too often rebound after initially responding to targeted therapies. A similar three‑pronged strategy, adjusted to each tumour type, could eventually shift treatment standards in those diseases as well.
The project has also highlighted the role of sustained public and charitable funding. Support from organisations such as the CRIS Foundation against cancer and the European Research Council allowed the team to pursue a long, risky line of research that might not have attracted immediate commercial investment.
What patients and families should realistically expect
For anyone facing pancreatic cancer today, this news does not change treatment tomorrow morning. Standard options remain surgery when possible, followed by combinations of chemotherapy, sometimes with radiotherapy, and participation in clinical trials where available.
Where the Spanish work does make a difference is in the outlook for the coming decade. It shows that even in a disease stuck in a stalemate for nearly fifty years, new thinking can produce fresh leads. Instead of hoping for one “magic bullet”, researchers are now designing coordinated attacks that hit cancer cells from several angles at once.
Rather than a miracle cure around the corner, this research points to a slow but real shift towards smarter, layered treatments for hard-to-treat tumours.
Key concepts worth unpacking
What is KRAS, in plain language?
KRAS is a gene that gives instructions for making a small protein acting like a switch. In healthy cells, that switch turns growth signals on and off in response to the body’s needs. In many cancers, KRAS is mutated so that the switch gets jammed in the “on” position.
Once that happens, cells receive a constant “grow and divide” order, even when they should rest or die. Targeted drugs try to either fix the switch or block the messages it sends further down the line.
Why are combinations often more powerful than single drugs?
Cancer cells are genetically unstable. When a single pathway is blocked, they can sometimes survive by using backup routes or by acquiring new mutations. Using combination therapies makes this adaptation harder.
There is a trade-off. Each added drug brings extra risks of side effects. The challenge is to find a combination where the benefit to patients clearly outweighs the additional toxicity. That is why careful, stepwise clinical trials remain unavoidable.
What the future might look like for patients
If future human trials of similar three‑drug regimens show promising results, pancreatic cancer care could start to look different. Instead of one-size-fits-all chemotherapy, patients might receive treatments tailored to the exact mutations in their tumour and the pathways those mutations activate.
In practice, that could involve genetic profiling of each tumour, followed by a personalised “cocktail” of targeted agents and, where needed, chemotherapy or immunotherapy. Responses would be tracked closely with scans and blood tests, adjusting combinations as tumours change over time.
For now, the Spanish breakthrough stands as a proof of concept: when researchers hit pancreatic cancer hard, in the right places and at the same time, even this stubborn disease can be forced into retreat in the lab. The next challenge is seeing how much of that success can safely cross from mice to people.